However, as water continues to cool below 4 degrees Celsius, the behavior of the water molecules changes again due to the formation of hydrogen bonds. As water molecules begin to cool, they lose energy and their motion slows down, allowing the attractive forces between them to pull them closer together, thereby increasing their density. 
At this temperature, the water molecules are more closely packed together due to the fact that the intermolecular forces between them are stronger. 
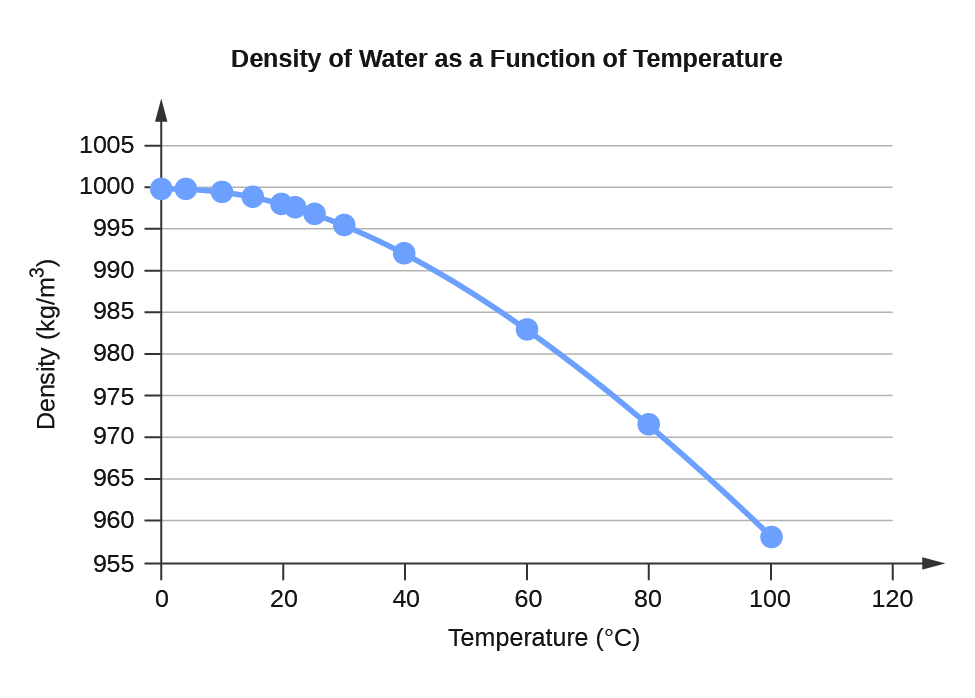
Water is more dense at 4 degrees Celsius due to the unique behavior of water molecules. Why is water more dense at 4 degrees Celsius? In wastewater treatment, the density of water is used to determine the optimal aeration rate for biological treatment, and in drinking water treatment, it is used to determine the optimal coagulant dosage required for removing suspended particles from water. In geophysics, the density of water is used to study groundwater resources and to determine the porosity and permeability of aquifers. In hydrodynamics, the study of fluids in motion, water density is used to determine the velocity and direction of water flow in rivers, lakes, and other bodies of water. The density of water has many practical applications in science, engineering, and industry. The relationship between the density of water and its salinity is often used in oceanography and other fields to study ocean currents and other phenomena. The saltwater’s density varies depending on the amount of dissolved salts and minerals present in the water. Salts and other dissolved substances in water increase its density, while freshwater has a lower density. The salinity of water also affects its density. Similarly, as the pressure increases, the density of water also increases, while as the pressure decreases, the density decreases. This is because the water molecules’ arrangement becomes less ordered, and the density decreases. As the temperature of water increases or decreases, its density decreases. As mentioned earlier, water has its maximum density at 4 degrees Celsius. The density of water is influenced by several factors, including temperature, pressure, and salinity. 
Density meters use various techniques, such as oscillation or pressure, to measure the density of water and other liquids accurately. Pycnometers are containers with a known volume that are filled with water and weighed to determine the density. When placed in water, the hydrometer floats, and the level of submersion indicates the water’s density. A hydrometer is a device that consists of a cylindrical glass tube with a weighted bulb at the bottom. There are several methods for measuring the density of water, including hydrometers, pycnometers, and density meters. This is the most commonly used reference point for the density of water. The density of water at 4 degrees Celsius in kg/m³ is 1000 kg/m³. What is the density of water at 4 degree Celsius in kg m3? The density of other substances is often compared to water to determine their relative buoyancy, solubility, and other properties. For example, the density of ice is less than that of liquid water at 4 degrees Celsius, which is why ice floats on water. The density of water at 4 degrees Celsius is often used as a benchmark for the density of other liquids and solids. The value of 1000 kg/m³ or 1 g/cm³ is the most commonly used reference point for the density of water at 4 degrees Celsius. These are some of the common units used to express the density of water, and their equivalents in the SI (International System of Units) and the English Engineering system. This is due to the water molecules’ arrangement, which is in a highly ordered and dense structure. At this temperature, water has its maximum density, which means that it occupies the smallest volume for a given mass. This value is often used as a reference point for the density of other substances. The density of water at a temperature of 4 degrees Celsius is 1000 kilograms per cubic meter (kg/m³) or 1 gram per cubic centimeter (g/cm³). In this blog post, we will focus on the density of water at a temperature of 4 degrees Celsius. The density of water changes with temperature, pressure, and salinity. One of the most important properties of water is its density, which is the mass of water per unit volume. Water is an essential element of life, and its properties play a vital role in many scientific and industrial applications. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
